Nuclear Magnetic Resonance (NMR) spectroscopy is the principle method of analysis for the characterisation and identification of organic compounds.
Typical applications of solution-state NMR methods include:
- Quantitative analysis of the content and purity of a sample
- Determination of molecular structure of organic molecules using 1H and 13C atom focused methods to identify the carbon-hydrogen framework of an organic compound
- Identification of molecular conformations
- Studies of physical and chemical properties at the molecular level such as conformational exchange and phase transitions
- Monitoring chemical processes and kinetics
- Studies of aggregation of molecules and diffusion
When applied to proteins, NMR spectroscopy allows the determination of protein structures, study of dynamics from ps to second, and study of interactions with other proteins or small molecule ligands in solution. Other directions of research in this area include analysis of carbohydrates and polymers in solution.
The main capabilities of protein NMR in Norwich include:
- Calculation of the structure of proteins in solution, using distance and torsion angle restraints, and other parameters measured by NMR
- Study of the dynamics of proteins in solution by measuring relaxation parameters that report on motions on the ps-ns timescale, and relaxation dispersion to report on dynamical processes in the ms timescale. This can be combined with hydrogen/deuterium exchange experiments to reveal how stable hydrogen bonds within the protein are and measure dynamics in a much longer timescale.
- Study of interactions of proteins with small or large molecules by NMR. NMR reports on individual atoms, and is therefore able to map each amino acid residue within the protein that is affected by each interaction.
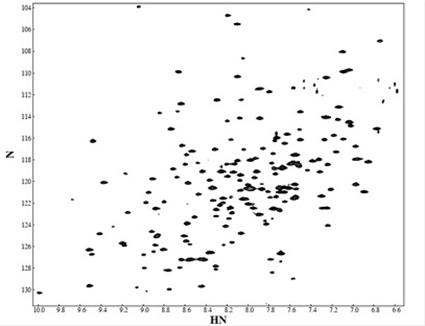
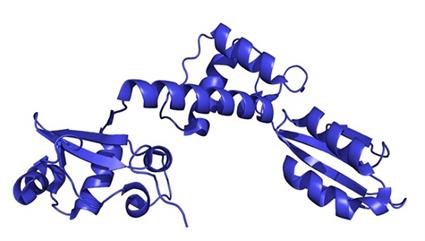
A [1H, 15N}-TROSY (transverse relaxation optimised spectroscopy) spectrum of the protein SBDS (above)) gives its signature pattern, a 29-kDa protein which is mutated in Shwachman-Bodian-Diamond syndrome, and the ribbon diagram structure of SBDS.
unt.
Solid-state NMR spectroscopy has proven to be an important tool capable of deriving structural and dynamics information using relatively short-range magnetic interactions.
To derive such information this technique has to overcome the effect of anisotropic magnetic interactions which, although containing a wealth of structural information, lead to significant broadening of the peaks, often beyond the expected level of resolution. The prevalent approach for eliminating this problem lies in the application of magic angle spinning. This method is suitable not only for solids, but also for colloidal materials and gels.
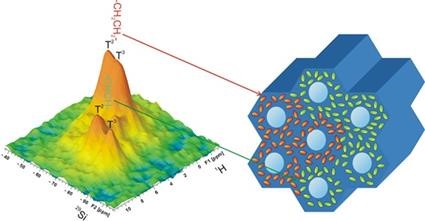
1H-29Si HETCOR solid-state NMR spectrum of periodic mesoporous organosilicas proving heterogeneous distribution of functional groups in amorphous framework
We have developed research expertise in elucidation of structural and dynamic properties of different classes of advanced materials:
- porous solids (zeolites, mesoporous silicas and polymers, metal-organic frameworks)
- polymer based amorphous pharmaceutical dispersions
- air and water sensitive materials
- pharmaceuticals and composites
We also have a variable temperature capability and have expertise in in-situ solid-state NMR.
Study soft materials and gels
In addition to solid-state NMR capability, the NMR facility at UEA provides a unique opportunity to study soft materials and gels. The application of either traditional solution or solid-state NMR methods to such systems is limited and therefore the use of magic-angle spinning in combination with advanced high-resolution methods is required. The systems of interest in this area include hydrogels, liquid-solid composites, gels, emulsions and tissues. The experiments on such systems can be carried out both at 400 and 800 MHz.

)